CORRA - Kinetic investigation of gasification reactions of chars from biomass
Motivation
Fuels on a biomass basis have the advantage that they can substitute fossil fuels without a change of infrastructure and can give an important contribution to the application of renewable energies. In contrast to the hydrogen technology, which is assumed as fuel of the future, synthetic fuels from biomass can directly be used by current engines and give the opportunity for improvements of engine design and efficiency. Therefore, the Forschungszentrum Karlsruhe GmbH proposes a new two step process to gasify fast growing biomass, i.e. cereal straw, hay and other organic residues. First, pyrolysis oil and char is produced in a fast pyrolysis plant. While preserving most of the energy content a volume reduction of 90 % is obtained. It is intended to mix pyrolysis oil and char to get slurry. Together with char the energy containment is enhanced to about more than a half of fossil oil. This slurry can easily be transported from several local pyrolysis plants to a central entrained flow gasifier of at least 500 MW thermal power. In a second process step the slurry is converted to a tar free syngas using entrained flow gasifiers. This step has successfully been tested in a 5 MW entrained gasifier at Future Energy in Freiberg/Sachsen, using pyrolysis oil and coke of beech wood.
Since, the physical and chemical processes during the gasification of a biomass slurry are not yet well understood, different accompanying experiments are performed, for instance gasification in an atmospheric gasifier with optical insight for spectral methods in order to determine velocity and concentration distributions for an accompanying numerical simulation. For this purpose a kinetic model is need to couple fluid dymanics with relevant chemical reactions in during a gasification process. These investigations will focus on the gasification behaviour of char particles in a CO2 and H2O rich environment, since a good understanding of char reactivity makes it possible to improve gasifier design and efficiency.
Problem
During char gasification the following main reactions may be considered:
| C + CO2 -->> 2 CO |
ƒ´H = +159,7 kJ/mol |
(1) |
| C + H2O -->> CO + H2 |
ƒ´H = +118,9 kJ/mol |
(2) |
| C + O2 -->> CO2 |
ƒ´H = -405,9 kJ/mol |
(3) |
| CO +H2O -->> CO2+H2 |
ƒ´H = -40,9 kJ/mol |
(4) |
| C + 2 H2 -->> CH4 |
ƒ´H = -87,4 kJ/mol |
(5) |
Reactions (1) and (2) are endothermic and can be considered as the most important in a gasification process. Oxidation reaction (3) provides the energy needed for the promotion of reactions (1) and (2). The resulting temperature level will adjust the chemical equilibrium in reaction (4) and will determine the syngas concentration and its qualitiy concerning further synthesis reactions. Therefore, the focus of this paper will be on reaction (1) and (2). Commercial char from beech wood as well as char from wheat straw and rice straw is investigated, the apparent kinetic data for reaction (1) and (2) are determined and compared to the coke of Columbian coal type 4. Additonally the problem of transport limitations is eluminated.
CORRA-Reaktor
The CORRA-reactor (Char Reaction Rate Acquisition-Reactor) is a fixed bed reactor in which the char is isothermally gasified with CO2 or H2O/N2 at temperatures between 825-1100 ¢XC. These low temperatures are chosen to exclude diffusion controlled limitations. The experimental set up is shown in Fig 1. The complete char sample of about 3 g is fed at once into the reactor from top with transport CO2 or H2O/N2. The product gas leaving the reactor is analysed by FID and a CO/CO2 analyser. In case of the CO2 reaction measurements are performed until CO production is determined. From the measured CO profile the conversion of char in dependence of time is determined, from which the best kinetic model is chosen (homogeneous-reacting core, non reacted core-shrinking core, modified volumetric model). In case of H2O gasification reactions the sum of CO and CO2 is analysed in order to consider the subsequent water gas shift reaction (4). Different fractions of particle sizes (0-25 µm, 25-40 µm, 50-80 µm, 80-100 µm and 100-200 µm) are investigated in order to find out if transport limitations during this heterogeneous reaction takes place or if the CO2 gasification reaction is controlled chemical kinetics.
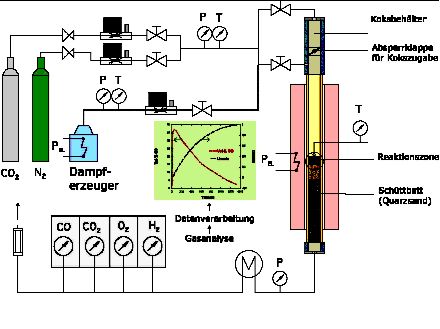 |
| Fig. 1 Experimental set-up of the CORRA reactor |
Results
Concerning the reaction C + CO2 -->> 2 CO for chars from wheat straw and beech wood the non reacted core model is favoured, where as in case of coke from coal the homogeneous model is best. The CO2 reactivity of chars from wheat straw is comparable. The determined apparent activation energies of of both reaction are similar (char rom beech wood: Ea = 144 kJ/mol, ln (A/s) = 5,9, char from wheat straw: Ea = 143 kJ/mol, ln (A/s) = 6,3). Investigation of CO2 reactivity of char from rice straw shoh a slightly higher reactivity: Ea = 133 kJ/mol und präexponentiellen Faktor ln (A/s) = 2,3. In case of coke from coal much higher apparent activation energies are determined, i.e. Ea = 214 kJ/mol, ln (A/s) = 4,9. The reaction C + H2O -->> CO + H2 is recently in focus of our investigation.
